FICS Product
FICS Attest
GxP validation in days, not months. AI-generated documentation, automated traceability, and native 21 CFR Part 11 compliance.
What It Is
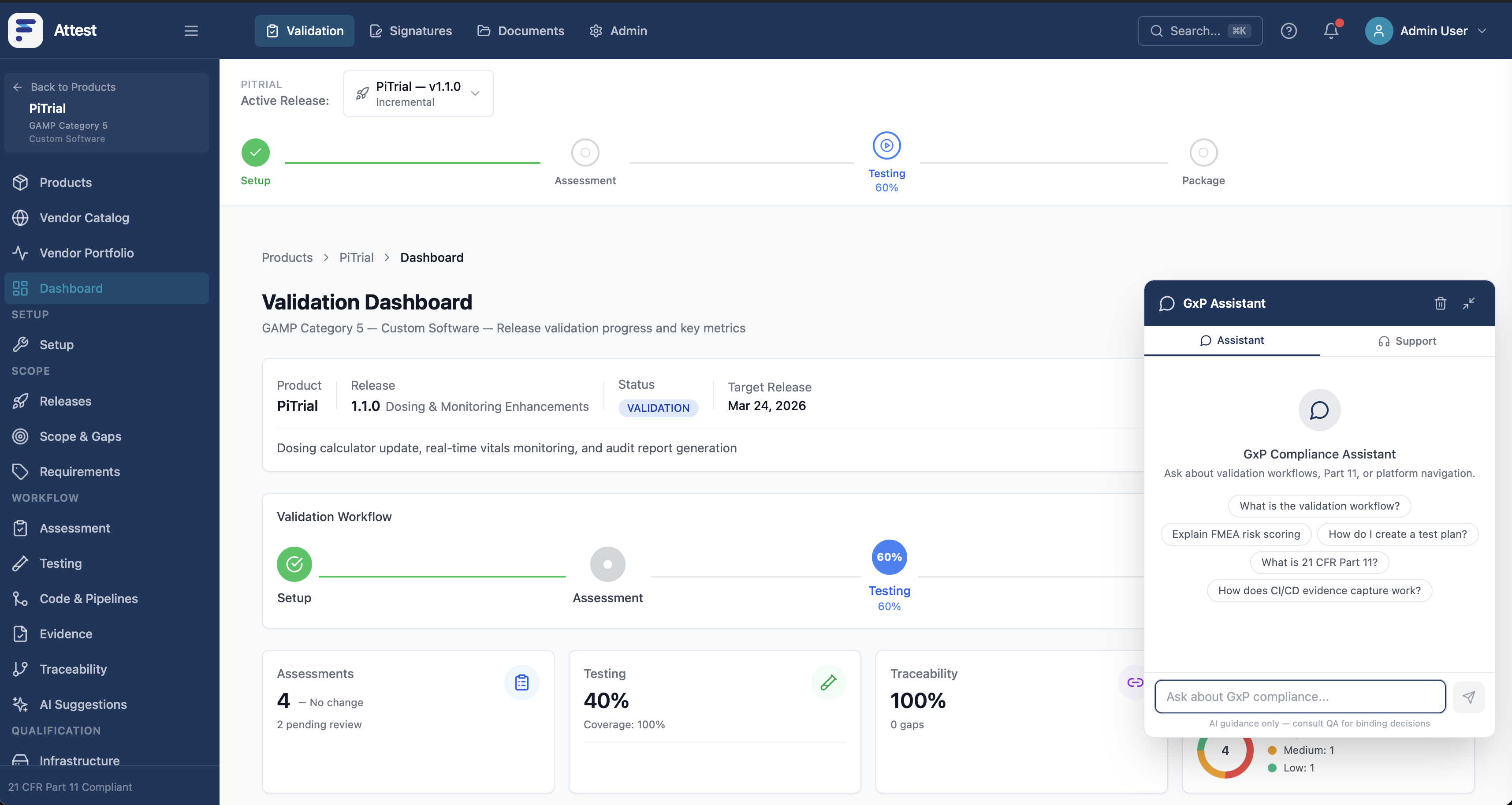
FICS Attest is an AI-powered validation tool that transforms the GxP validation lifecycle. Traditional validation cycles take weeks or months and rely on manual documentation across fragmented systems. FICS Attest auto-generates validation documentation, executes traceability mapping, and maintains 21 CFR Part 11 audit trails natively — consolidating 4-5 external vendor tools into a single in-house platform.
Who It's For
Quality assurance teams, validation engineers, compliance officers, and IT teams responsible for maintaining GxP-compliant systems in clinical trial environments.
Why FICS
Why It's Better with FICS
Reduces validation time from weeks to days with AI-generated documentation
Eliminates manual compliance overhead — traceability mapping is automated
Native 21 CFR Part 11 audit trails — no separate e-signature or audit trail tools needed
Consolidates eQMS, e-signatures, validation lifecycle, and audit trail into one platform
Saves $150K+ in annual vendor spend by replacing 4-5 external tools
Continuous validation — changes are automatically re-validated, not batch-processed quarterly
Ready for Clinical Development 2.0?
Partner with Pi Health and experience clinical trials reimagined: smarter, faster, and globally scalable.
Request a Demo