FICS Product
eTMF
An electronic Trial Master File that maintains itself. Documents flow in automatically, filed correctly, audit-ready from day one.
What It Is
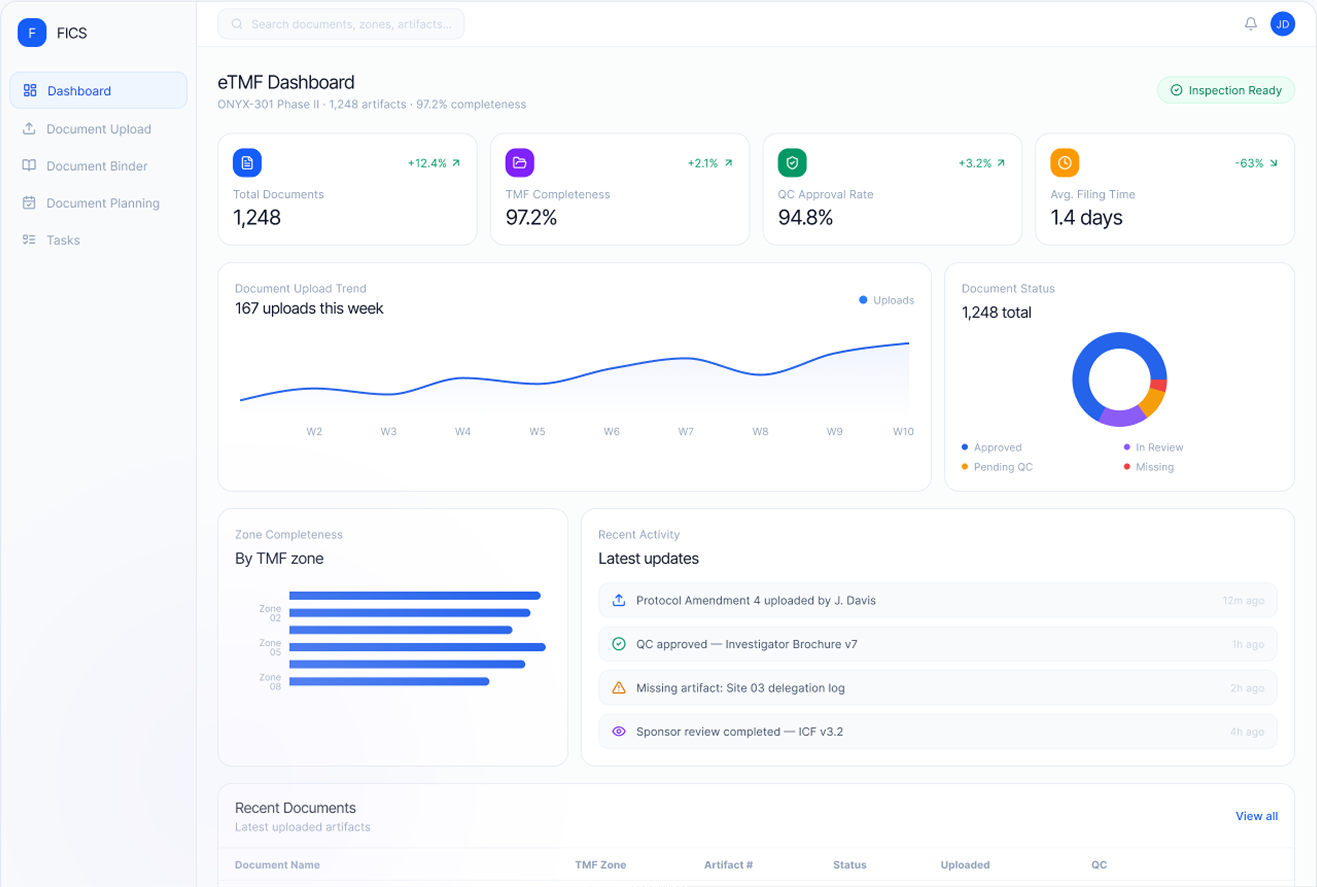
FICS eTMF is a fully integrated electronic Trial Master File that eliminates manual document filing and classification. As trial activities occur across CTMS, EDC, and safety workflows, the relevant documents are automatically generated, classified, and filed into the correct TMF structure — maintaining inspection readiness at all times.
Who It's For
Regulatory affairs teams, quality assurance, TMF managers, and clinical operations teams who need a perpetually inspection-ready Trial Master File without the manual overhead.
Why FICS
Why It's Better with FICS
Automated filing — documents generated from trial workflows are filed in seconds, not days
Always inspection-ready — completeness dashboards show real-time TMF health
Unified with ISF — investigator site files stay synchronized with the sponsor TMF automatically
No reconciliation — because documents originate from FICS workflows, there is no gap between source and TMF
AI-powered document classification and quality checks
Sponsor document exchange built in — streamlined collaboration between sponsors and sites
Ready for Clinical Development 2.0?
Partner with Pi Health and experience clinical trials reimagined: smarter, faster, and globally scalable.
Request a Demo