FICS Product
CTMS
Clinical Trial Management, reimagined. A workflow-driven CTMS built natively into the FICS operating system.
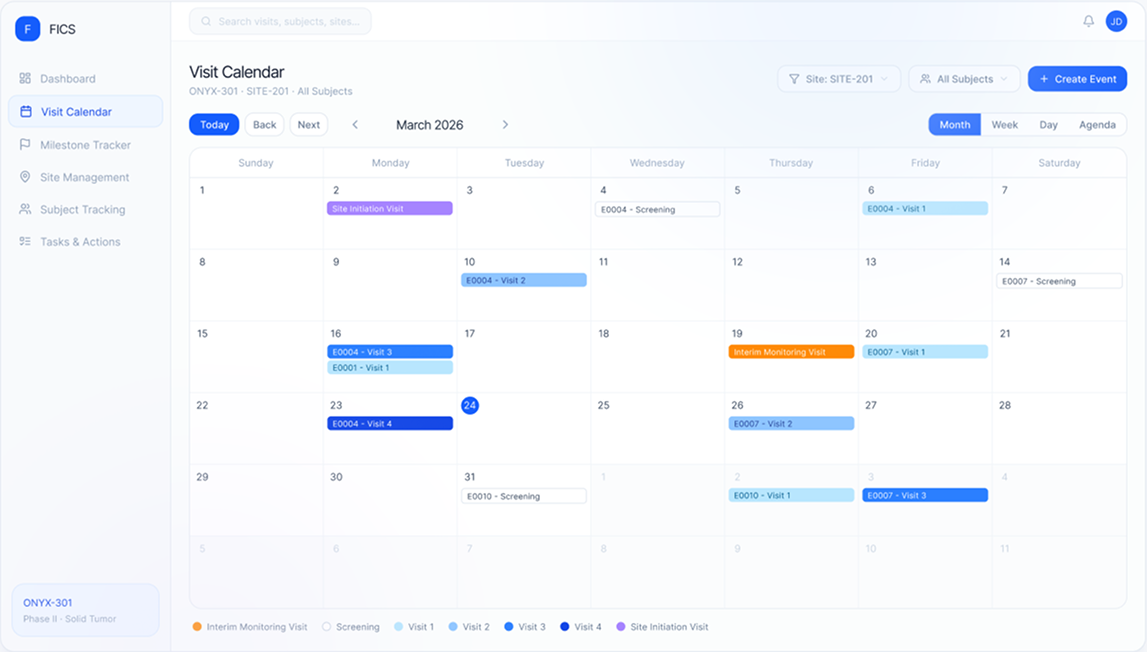
What It Is
FICS CTMS is a next-generation clinical trial management system that unifies study planning, site management, milestone tracking, and operational oversight into a single workflow-driven platform. Unlike traditional CTMS tools that operate as standalone databases, FICS CTMS is deeply integrated with EDC, eTMF, and safety — eliminating data silos and manual reconciliation.
Who It's For
Clinical operations teams, project managers, CTMs, and sponsor oversight teams who need real-time visibility into trial progress without toggling between disconnected systems.
Why FICS
Why It's Better with FICS
Unified data layer — site, subject, and visit data flows automatically across CTMS, EDC, and eTMF with zero reconciliation
Workflow-driven execution — milestones, tasks, and escalations are triggered by real trial events, not manual updates
Real-time sponsor and site visibility — live dashboards reflect actual study state, not stale exports
AI-powered analytics — predictive enrollment modeling and risk-based monitoring built in
Amendments propagate automatically across all connected workflows
Single audit trail across the entire trial lifecycle
Ready for Clinical Development 2.0?
Partner with Pi Health and experience clinical trials reimagined: smarter, faster, and globally scalable.
Request a Demo